Post #4 in a series from students working in the Wyeth lab this summer. This time it’s Victoria Tweedie-Pitre and Lauren PIctou working with Yulia Reunova on gastropod neuroanatomy.
More from Victoria and Lauren…
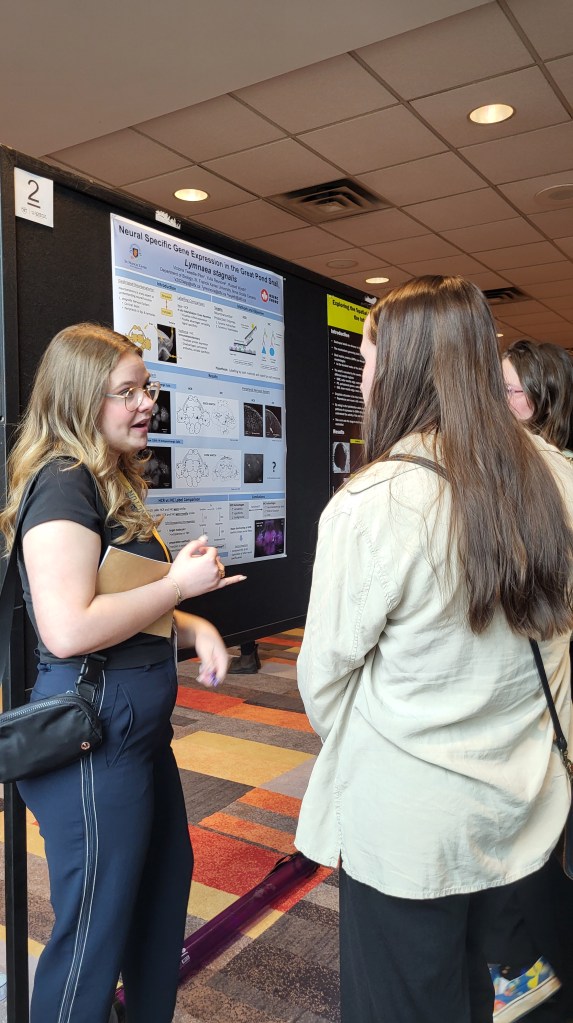
The Wyeth Lab neuroanatomy team is currently focused on identifying and describing different neuron types in the nervous system of the great pond snail, Lymnaea stagnalis. This freshwater pulmonate is commonly used as a model organism in neurobiology due to its relatively large (including giant neurons!) and fewer neurons making it ideal for detailed studies.
Neuroanatomy is a crucial starting point in neuroethology, as mapping out the structural framework of neurons and their connections is essential before understanding how the nervous system controls behavior. By using genes and functional proteins associated with neurotransmitter synthesis, we can classify different neuron types. Our research aims to advance neuronal classification by investigating the anatomical morphology and distribution patterns of neurons containing various neural-specific genes and proteins in L. stagnalis.
We use two methods: immunohistochemistry and in situ hybridization chain reaction (HCR). Immunohistochemistry is a protein-based technique that uses antibodies to label antigens, while HCR is an mRNA-based technique that labels gene target sequences. Recently, we have been combining both techniques in a single protocol, allowing us to closely visualize neurons that contain both mRNA and the functional proteins involved in neurotransmitter synthesis.
Our current investigations focus on several key enzymes involved in neurotransmitter synthesis:
- Tyrosine Hydroxylase (involved in dopamine synthesis)
- Dopamine Beta-Hydroxylase (involved in norepinephrine synthesis)
- Tyramine Beta-Hydroxylase (involved in octopamine synthesis)
- Choline Acetyltransferase (involved in acetylcholine synthesis)
By exploring these enzymes with different molecular techniques, we aim to uncover new insights into the complex organization and function of the nervous system in L. stagnalis. Overall, this will help bring us a step closer towards the goal of understanding how the nervous system works to control behaviour!